Development Pipeline
Tempest is developing a first-in-class oncology pipeline of oral therapeutics with broad potential for patients. The product candidates are designed to treat cancer by direct tumor killing, activating the immune system to kill tumor cells, or a combination of both mechanisms.
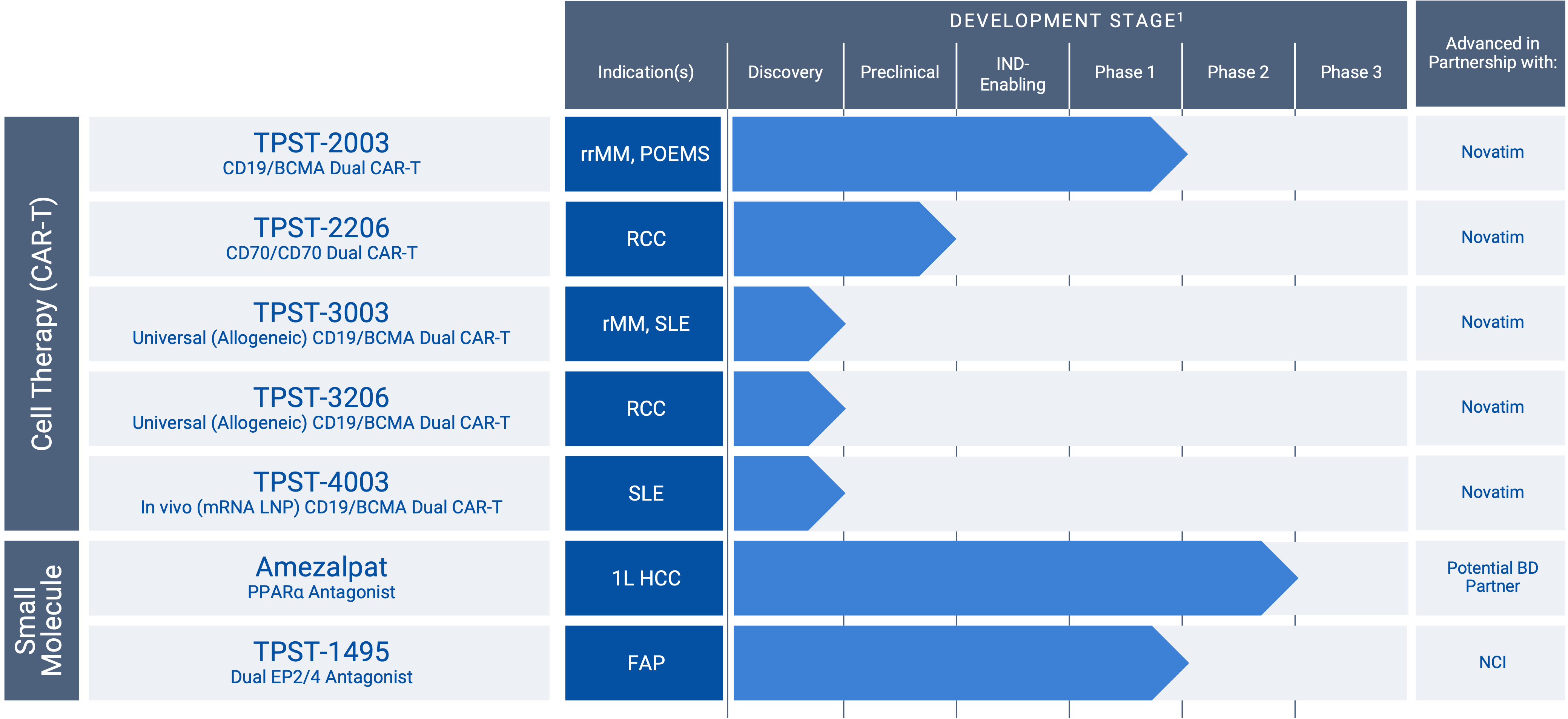

1For amezalpat, Phase 3 timelines are subject to a partnership and/or separate funding. TPST-1495 Phase 2 to be operationalized by the Cancer Prevention Network of the National Cancer Institute (“NCI”). TPST-2003, TPST-2206, TPST-3003, TPST-3206, and TPST-4003 clinical development in China to be operationalized by Novatim Immune Therapeutics (Zhejiang) Co., Ltd.
“RCC” renal cancer; “HCC” hepatocellular carcinoma; “CCA” cholangiocarcinoma; “FAP” familial adenomatous polyposis; “FPI” First Patient In; “rrMM” relapsed/refractor multiple myeloma, “SLE” lupus.
